Trending...
- Susan Keel Named President of Metro Builders Association
- Acquisition of Israeli Defense Manufacturing Platform to Accelerate AI-Driven Autonomous Systems: VisionWave Holdings, Inc.: (N A S D A Q: VWAV)
- 106 Years Strong: The Liberty Group Celebrates a Century-Plus of Service and Unveils a Unified Family of Companies
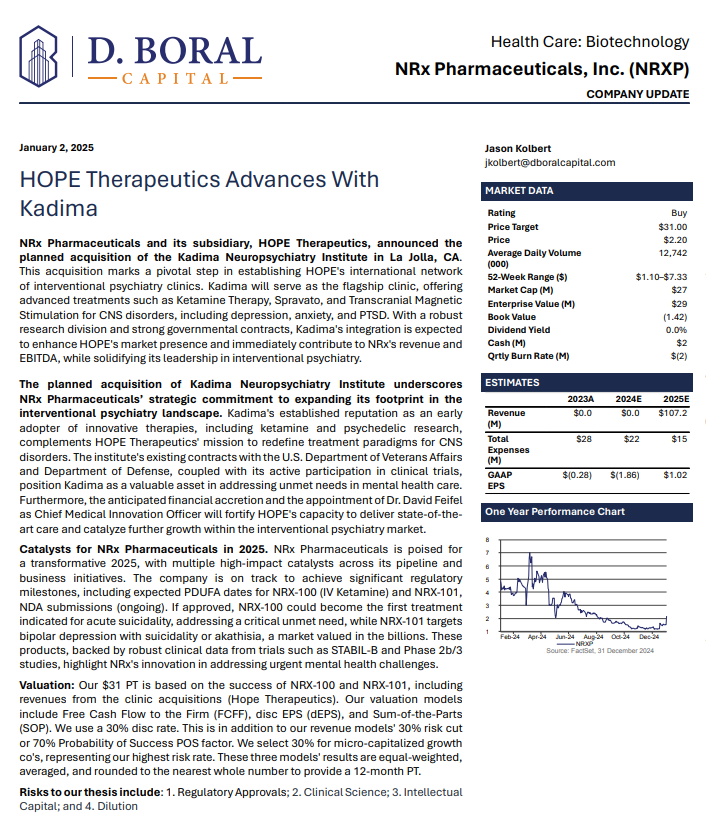
$NRXP given a $31 Price Target from Respected Investment Analyst D. Boral Capital
MIAMI - WisconsinEagle -- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
$27 Million Funding Transaction Signed for HOPE Clinic Acquisition.
Intent to Initiate Nationwide Acquisition Program in Partnership with Leading Investment Bank in Early February.
TV Feature on Sanjay Gupta/CNN Program Covering Ketamine with Kadima Neuropsychiatry Institute Founder Dr. David Feifel, Expected to Join NRXP.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
Conducted Investor Workshop to Support Planned Expansion of Subsidiary HOPE's Network at the Mar-A-Lago Club in Palm Beach, FL.
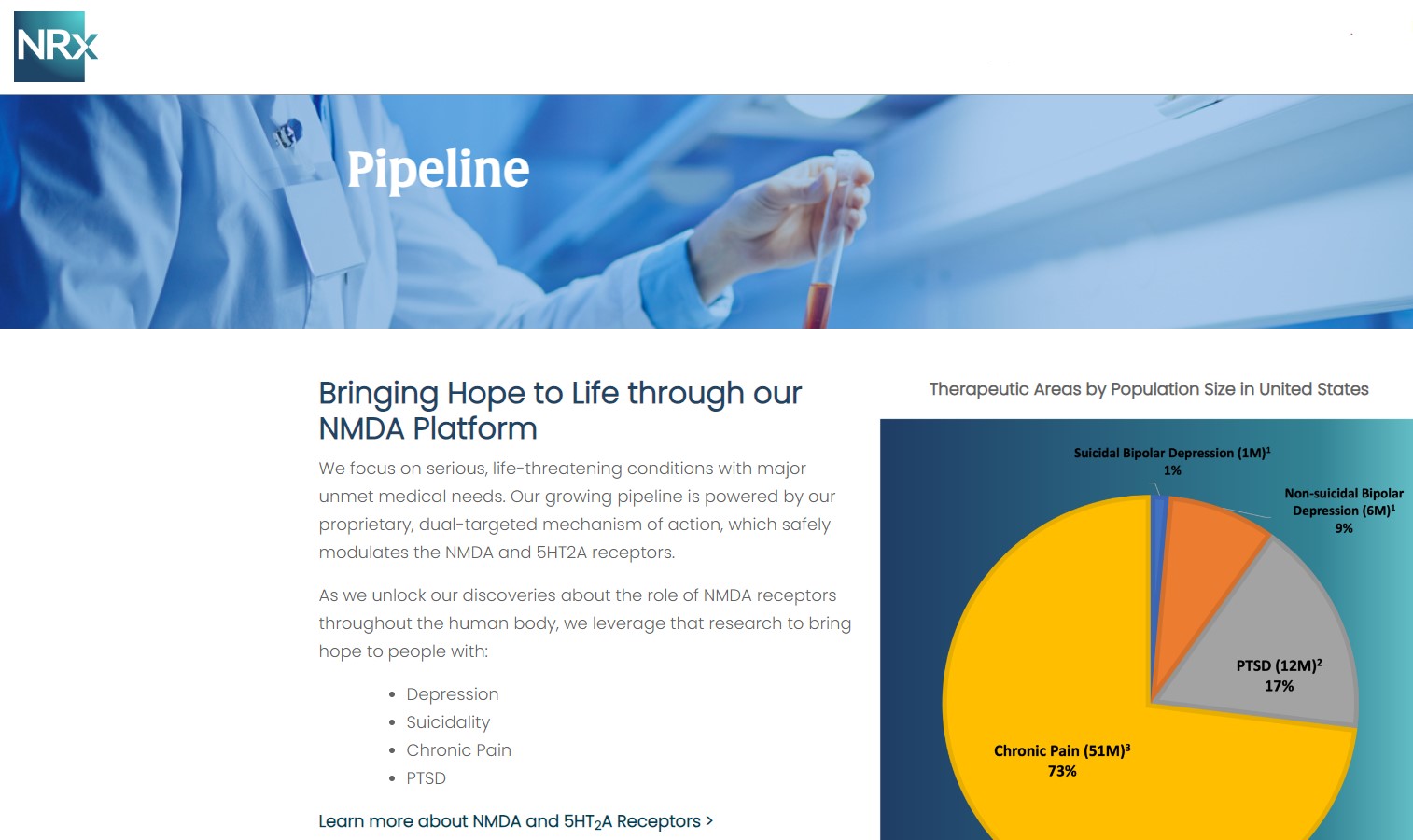
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
More on Wisconsin Eagle
In a January 2025 report, respected investment analysis firm D. Boral Capital assigned NRXP a $31 Price Target. The full report may be viewed at this link: https://www.nrxpharma.com/wp-content/uploads/2025/01/HOPE-Therapeutics-Advances-With-Kadima.pdf.
$27 Million Funding Transaction for HOPE Clinic Acquisition
On January 28th NRXP announced the signing and of a Stock Purchase Agreement with Smith & Sauer, LLC, as the first step in a $27 million financing to fund the planned acquisition of HOPE clinics. The agreement provides for the immediate funding of $2.0 million in NRXP common stock and commits Smith and Sauer to purchase an additional $25 million in newly issued Series A Preferred Stock of HOPE, the proceeds of which are intended to be used to initiate a nationwide rollup of Interventional Psychiatry Clinics to treat patients with severe, suicidal depression and PTSD. The first $6.75 million tranche is committed by February 7, 2025, with the full $25 million committed on or before April 1, 2025.
The Company anticipates leveraging the Smith and Sauer investment with bank debt and similar instruments to fund an initial rollup of Interventional Psychiatry Clinics with anticipated revenues of approximately $100 million on a pro-forma basis by the end of 2025. Unlike "ketamine clinics" that are frequently operated by non-mental health professionals, NRXP HOPE anticipates building a best-in-class nationwide chain of facilities that combine treatment with ketamine and other psychedelic medications with Transcranial Magnetic Stimulation (TMS) and recently FDA-approved Digital Therapeutics to best serve patients with life-threatening depression, PTSD, and related disorders.
More on Wisconsin Eagle
Sanjay Gupta/CNN Program on Ketamine, Featuring Kadima Neuropsychiatry Institute Founder Dr. David Feifel, Expected to Join NRXP
On January 14th NRXP alerted investors that Kadima Neuropsychiatry Institute's Founder David Feifel, MD PHD was featured as an expert in a program on ketamine entitled "'Special K': The Science & Stigma of Ketamine" on CNN, hosted by Dr. Sanjay Gupta. NRXP previously announced that Kadima is identified as the Company's planned flagship clinic acquisition, upon closing of the transaction.
Dr. Feifel, a global leader in interventional psychiatry, has accepted a future role as Chief Medical Innovation Officer in conjunction with the previously announced planned acquisition of Kadima by NRXP.
In his comments, Dr. Feifel supported use the of ketamine in appropriate patients but emphasized the need for appropriate psychiatric supervision in a controlled setting - a view that NRXP wholeheartedly shares. This appearance with Dr. Gupta follows his appearance on another of Dr. Gupta's programs, "The Wild West of Ketamine Treatment" in August of last year.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
On January 13th NRXP announced it has applied to NASDAQ to uplist the Company's securities to the NASDAQ Global Market (NASDAQ GM).
Listing on the NASDAQ Global Market requires NRXP to meet published requirements of the NASDAQ Global Market, including maintaining a market value of listed securities (MVLS) above $75 million and a minimum bid price of at least $4.00 for 90 consecutive trading days. NRXP has advised NASDAQ that it currently has 15.7 million outstanding shares of common stock, which will inform the calculation of the Company's market capitalization.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
$27 Million Funding Transaction Signed for HOPE Clinic Acquisition.
Intent to Initiate Nationwide Acquisition Program in Partnership with Leading Investment Bank in Early February.
TV Feature on Sanjay Gupta/CNN Program Covering Ketamine with Kadima Neuropsychiatry Institute Founder Dr. David Feifel, Expected to Join NRXP.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
Conducted Investor Workshop to Support Planned Expansion of Subsidiary HOPE's Network at the Mar-A-Lago Club in Palm Beach, FL.
NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year. This represents a $3-5 billion market at expected pricing. Based on the data in the trials referenced above, the Company's regulatory counsel encouraged the Company to file an NDA for suicidal depression for NRX-100.
More on Wisconsin Eagle
- The State of Law Firm Marketing: Top Companies, Awards, and Resources
- USA Best Book Awards Finalist What Love Leaves Behind Releases March 24
- Inkdnylon Custom Apparel Launches Cost-Saving System for Promotional Products and Custom Apparel in Chicago
- ENTOUCH Named Finalist for 2026 North American Inspiring Workplaces Awards
- Cleveland County Goat Farm NC Kikos Featured in "Feature Farmer Friday" Documentary
In a January 2025 report, respected investment analysis firm D. Boral Capital assigned NRXP a $31 Price Target. The full report may be viewed at this link: https://www.nrxpharma.com/wp-content/uploads/2025/01/HOPE-Therapeutics-Advances-With-Kadima.pdf.
$27 Million Funding Transaction for HOPE Clinic Acquisition
- $25 million investment in HOPE to consist of Series A (non-dilutive to NRx shareholders) convertible into 1/3 of fully diluted HOPE equity, with a 15% current preferred dividend (non-callable for 2 years) for planned HOPE clinic acquisitions
- Initial funding of $2.0 million for purchase of NRx common stock anticipated to close on or before January 29, with the first tranche of $6.75 million of $25.0 million investment to be invested in HOPE Therapeutics, Inc. on or before February 7, 2025, and the balance of $18.25 million by April 1, 2025
- Funds will be used in conjunction with anticipated bank financing to initiate nationwide rollup strategy of interventional psychiatry clinics, starting with Kadima Neuropsychiatry (La Jolla, CA)
- HOPE intends to initiate nationwide acquisition program in partnership with leading investment bank on or around February 1, 2025.
On January 28th NRXP announced the signing and of a Stock Purchase Agreement with Smith & Sauer, LLC, as the first step in a $27 million financing to fund the planned acquisition of HOPE clinics. The agreement provides for the immediate funding of $2.0 million in NRXP common stock and commits Smith and Sauer to purchase an additional $25 million in newly issued Series A Preferred Stock of HOPE, the proceeds of which are intended to be used to initiate a nationwide rollup of Interventional Psychiatry Clinics to treat patients with severe, suicidal depression and PTSD. The first $6.75 million tranche is committed by February 7, 2025, with the full $25 million committed on or before April 1, 2025.
The Company anticipates leveraging the Smith and Sauer investment with bank debt and similar instruments to fund an initial rollup of Interventional Psychiatry Clinics with anticipated revenues of approximately $100 million on a pro-forma basis by the end of 2025. Unlike "ketamine clinics" that are frequently operated by non-mental health professionals, NRXP HOPE anticipates building a best-in-class nationwide chain of facilities that combine treatment with ketamine and other psychedelic medications with Transcranial Magnetic Stimulation (TMS) and recently FDA-approved Digital Therapeutics to best serve patients with life-threatening depression, PTSD, and related disorders.
More on Wisconsin Eagle
- Tony Grundler Introduces Artificial Intelligence V.S. Avatar-Ian's
- Hollywood's Elite Gather at the Annual WOW Creations Oscars Gifting Suite at the Universal Hilton
- Where Were the Women? Reframing the Greek Revolution Through Contemporary Art
- JGCMGS Details Architecture to Safeguard Assets From Unauthorized Phishing Scams
- 21 Days: The Malta Deadline That Could Redraw the Finnish Online Casino Map
Sanjay Gupta/CNN Program on Ketamine, Featuring Kadima Neuropsychiatry Institute Founder Dr. David Feifel, Expected to Join NRXP
On January 14th NRXP alerted investors that Kadima Neuropsychiatry Institute's Founder David Feifel, MD PHD was featured as an expert in a program on ketamine entitled "'Special K': The Science & Stigma of Ketamine" on CNN, hosted by Dr. Sanjay Gupta. NRXP previously announced that Kadima is identified as the Company's planned flagship clinic acquisition, upon closing of the transaction.
Dr. Feifel, a global leader in interventional psychiatry, has accepted a future role as Chief Medical Innovation Officer in conjunction with the previously announced planned acquisition of Kadima by NRXP.
In his comments, Dr. Feifel supported use the of ketamine in appropriate patients but emphasized the need for appropriate psychiatric supervision in a controlled setting - a view that NRXP wholeheartedly shares. This appearance with Dr. Gupta follows his appearance on another of Dr. Gupta's programs, "The Wild West of Ketamine Treatment" in August of last year.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market
On January 13th NRXP announced it has applied to NASDAQ to uplist the Company's securities to the NASDAQ Global Market (NASDAQ GM).
Listing on the NASDAQ Global Market requires NRXP to meet published requirements of the NASDAQ Global Market, including maintaining a market value of listed securities (MVLS) above $75 million and a minimum bid price of at least $4.00 for 90 consecutive trading days. NRXP has advised NASDAQ that it currently has 15.7 million outstanding shares of common stock, which will inform the calculation of the Company's market capitalization.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (Stock Symbol: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on Wisconsin Eagle
- Ozz Metals Ltd Secures 1-Tonne Gold Offtake Agreement
- Jet Set: The Ultimate Coachella Afterparty
- Heritage at Manalapan Introduces New Single-Family Home Community in One of Monmouth County's Most Desirable Locations
- Compliant Workspace announces partnership with Blackpoint Cyber
- Michigan Homeowners Urged to Act on Rising Basement Waterproofing Needs Amid Severe Flood
- Event Solutions Enters New Era: Announces New Leadership
- Carlsbad Hotel Named Best of La Quinta Award Winner
- Scoop Social Co. Launches a New Era of Mobile Hospitality — One Truck, Two Experiences
- Record Sales Growth After Strategic Acquisitions; New Distribution Agreements for Established Premium Cigar Supplier: Green Leaf Innovations $GRLF
- R2 Copilot Addresses Critical Privacy Issues as Enterprise AI Spending and Security Incidents Rise
- Innovative Environmental Technologies Unveils New Website Featuring Free AI Tools for the Environmental Industry
- CCHR Warns: Psychiatric Diagnoses Without Biological Proof Now Used to Justify Euthanasia
- Impact Filtration Appoints Alejandro Sturniolo as Head of Sustainability to Engineer High-Performance, Water-Positive Infrastructure
- How Best Friends Turned Their Love of Boba Into a Wellness Brand
- 106 Years Strong: The Liberty Group Celebrates a Century-Plus of Service and Unveils a Unified Family of Companies
- Airport Transportation Reaches All Five Continents Through Global Transportation Partner Network
- Susan Keel Named President of Metro Builders Association
- Acquisition of Israeli Defense Manufacturing Platform to Accelerate AI-Driven Autonomous Systems: VisionWave Holdings, Inc.: (N A S D A Q: VWAV)
- HRC Fertility to Celebrate Grand Opening of New Beverly Hills Location During National Infertility Awareness Week
- AktieGo Publishes Editorial Feature Examining Decentralized Power Infrastructure and Hydrogen Energy Deployment