Trending...
- Global Families Turn to Young Travelers Concierge Amid Rising Safety Concerns in the U.S
- Don't Leave Your Body Behind: The New Children's Book Series Helping Kids Move, Feel, and Thrive
- Tampa-Based Digital Marketing Agency Launches New Website to Help Local Businesses Grow Online
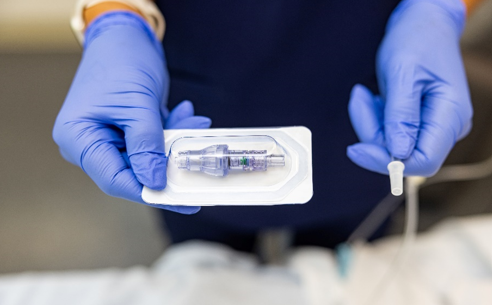
FAYETTEVILLE, Ark. - WisconsinEagle -- Lineus Medical® is proud to announce the U.S. Food and Drug Administration (FDA) has cleared its flag ship product, SafeBreak® Vascular, for use on all vascular access lines including peripheral IV catheters, midlines, peripherally inserted central catheters, central venous catheters, IV ports, and interosseous vascular access devices on patients two weeks of age and up. This allows SafeBreak Vascular to be installed on both adults and children and on every type of IV line.
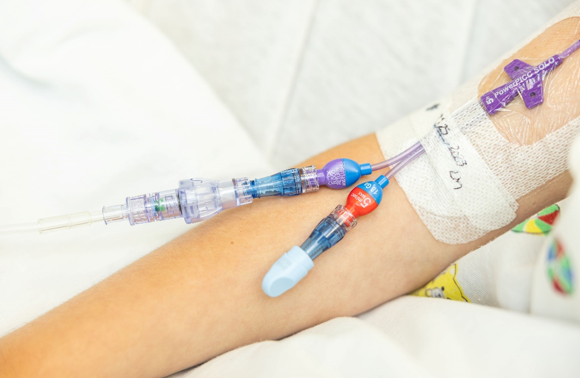
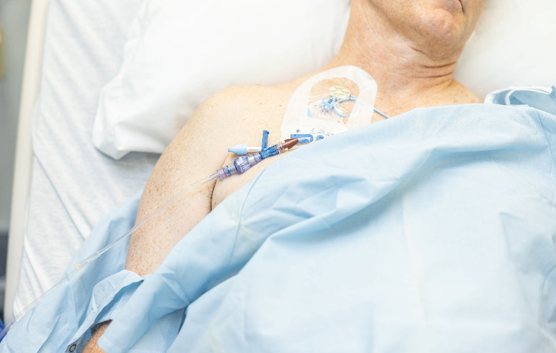
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on Wisconsin Eagle
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on Wisconsin Eagle
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on Wisconsin Eagle
- Robert Fabbio Inducted into the Austin Technology Council Hall of Fame
- Cybersecurity is Protecting Your Personal Information and Your Portfolio
- L2 Aviation Celebrates Grand Opening of New Facility at Cincinnati/Northern Kentucky International Airport (CVG)
- Managing Summer Staffing Surges with Confidence: Why Name Badges Are a Must for Seasonal Success
- Visa Named Title Sponsor of Ascending Athletes' Business Owners Summits for NFL Entrepreneurs
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on Wisconsin Eagle
- The Paris Court of International Arbitration Elects Dr. John J. Maalouf as its New President
- $56.7 Million Announced in Q1 2025 with Revenue Growth and Progress Toward NASDAQ Uplisting for AI Marketing Company: IQSTEL, Inc. Stock Symbol: IQSTD
- When The Call Hits Home Podcast Features Tom Rizzo
- SAVVY MINING raised $500 million and launched BTC.XRP.DOGE cloud mining, increasing investors' returns by 30%
- New National Nonprofit Launches to Capture Firsthand Accounts of Adoption Stories
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
Source: Lineus Medical
Filed Under: Business
0 Comments
Latest on Wisconsin Eagle
- Bonelli Systems Expands Managed IT Services Nationwide, Leveraging Microsoft Azure Expertise
- $4.3 Million Patent Application Waiver Fee Granted by FDA on New Drug Application Fee for Treatment Addressing Suicidal Depression & PTSD: NRX Pharma
- Whistleblower Claims Dental Patient Deaths Likely Due to Book Ban
- xREnergy up as much as +3,094,634% on first day listed on the XRP Ledger. Ticker : $XRE
- Psychiatry's Legacy of Racism and Coercion Highlighted in Restraint Deaths
- New Book 'Cybersecurity Leadership' Guides SME Leaders to Make Smart, Strategic Security Decisions
- "Stop scrolling and start watching" - Beloved film recommendation site Criticker gets a major makeover
- Green Energy Solar Expands with New Offices in Port St. Lucie, West Palm Beach, and Orlando
- Events by Dubsdread Expands Services to The Venue at Lake Lily
- London Gala Emphasizes Trade, FDI and Ongoing Cooperation
- Sahit Muja Launches Trillion-Dollar Green Revolution with 10 Groundbreaking Cryptos
- Free and Low-Cost Dental Care Now Available in London Through the Canadian Dental Care Plan (CDCP)
- Garden State Gay Socials Turns One: 1st Birthday Celebration for Gay Men Who Want Real Connection
- Sea Fox® 368X "Extreme Edition" by Black Label Marine Group Celebrates 5 Years of the Commander Legacy
- CCHR Florida: Mental Health Awareness Month Open House
- Introducing LĪNA Universal Balm: A Luxury Multi-Use, Plant-Powered Solution for Skin & Hair
- UIFCA Revolutionizes Financial Education with Elite Team of Investment Experts
- Global Families Turn to Young Travelers Concierge Amid Rising Safety Concerns in the U.S
- Sober.Buzz the Sober Token : Ticker $BUZZ
- Divorce-Online Marks 25 Years Transforming Family Law Services in the UK